
CancerWatch Survey Achieves Record-Breaking 94% Response Rate Across Europe
We are proud to share that the CancerWatch survey phase has officially concluded, marking a significant step forward for the Joint Action.

We are proud to share that the CancerWatch survey phase has officially concluded, marking a significant step forward for the Joint Action.

Europe has set itself an ambitious goal: to reduce the cancer burden for patients, families, and health systems across the EU. Through Europe’s Beating Cancer Plan, billions of euros are being invested in prevention, early detection, treatment, and survivorship.

The subtasks survival (7.1.1) and prevalence (7.1.2) started with a kick-off meeting that was well-attended by the active participants and observers.
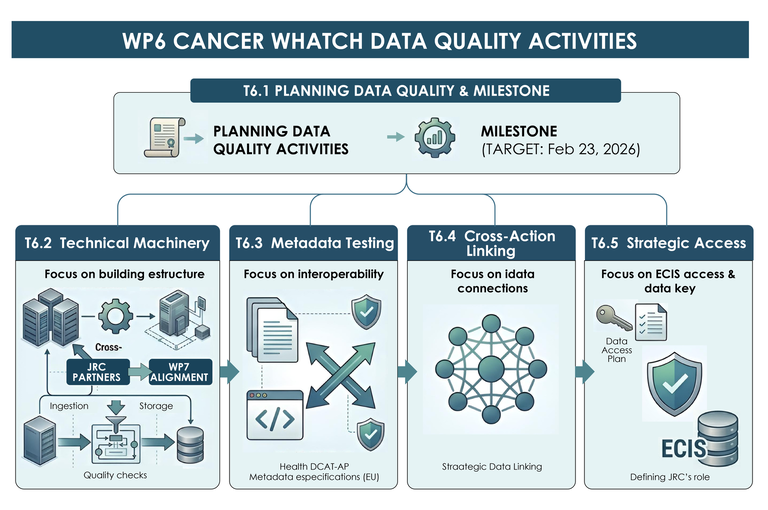
The kick-off online meeting of Task T6.1 was organized by the Foundation for the Promotion of Health and Biomedical Research (FISABIO), Valencia, Spain, with the participation of 27 attendees involved in the CancerWatch JA, the Joint Research Centre (JRC) in Ispra, and other data quality experts.

Our colleagues in Work Package 5, in close collaboration with the European Network of Cancer Registries (ENCR) and the Joint Research Centre (JRC), have officially launched a landmark survey on population-based cancer registry (PBCR) practices.

Last month the Committee, the Scientific Advisory Board (SAB) was formally established. On 6 February 2026, the project leadership met online with the board for the first time to kick off the collaboration and discuss priorities for the coming period.

We are delighted to share a glimpse of the 25th AIRTUM Scientific Meeting, the leading event for cancer registration and epidemiology in Italy!

Today in Oslo, CancerWatch officially kicks off a new EU Joint Action that will improve the quality, comparability, and timeliness of cancer data across Europe.

A strong endorsement of cancer registries from Oliver Varhelyi, the EU Commissioner for Health and Animal Welfare. “Data into decisions and decisions into impact”.